파고솜: 두 판 사이의 차이
"Phagosome" 문서를 번역하여 만듦 |
편집 요약 없음 태그: 2017 원본 편집 동음이의 링크 |
||
| 4번째 줄: | 4번째 줄: | ||
파고솜은 [[미생물]], [[세포 노화|노화]]된 세포, [[세포자살|세포자멸사]] 예정인 세포 주변의 [[세포막]]이 융합하여 형성된다. 파고솜은 [[리소좀]]을 동원하고 서로 융합하여 성숙한 [[파고리소좀]]을 형성하는 [[막 단백질]]을 가지고 있다. 리소좀에는 [[병원체|병원균]]을 죽이고 소화시키는 [[가수 분해 효소]]와 [[활성산소|활성 산소종]](ROS)이 포함되어 있다. 파고솜은 식작용 능력이 전문 식세포에 비해서 떨어지는 비전문 식세포에서도 형성될 수 있지만, 더 작은 범위의 입자만 삼킬 수 있으며 ROS는 들어 있지 않다. 소화된 입자에서 유용한 물질([[아미노산]] 등)은 [[세포기질|세포질]]로 이동하고 노폐물은 [[세포외 배출]]에 의해 제거된다. 파고솜 형성은 조직 [[항상성]]과 병원체에 대한 [[선천면역|선천성]], [[적응면역|적응성]] 면역에 중요하다. |
파고솜은 [[미생물]], [[세포 노화|노화]]된 세포, [[세포자살|세포자멸사]] 예정인 세포 주변의 [[세포막]]이 융합하여 형성된다. 파고솜은 [[리소좀]]을 동원하고 서로 융합하여 성숙한 [[파고리소좀]]을 형성하는 [[막 단백질]]을 가지고 있다. 리소좀에는 [[병원체|병원균]]을 죽이고 소화시키는 [[가수 분해 효소]]와 [[활성산소|활성 산소종]](ROS)이 포함되어 있다. 파고솜은 식작용 능력이 전문 식세포에 비해서 떨어지는 비전문 식세포에서도 형성될 수 있지만, 더 작은 범위의 입자만 삼킬 수 있으며 ROS는 들어 있지 않다. 소화된 입자에서 유용한 물질([[아미노산]] 등)은 [[세포기질|세포질]]로 이동하고 노폐물은 [[세포외 배출]]에 의해 제거된다. 파고솜 형성은 조직 [[항상성]]과 병원체에 대한 [[선천면역|선천성]], [[적응면역|적응성]] 면역에 중요하다. |
||
그러나 일부 [[세균]]은 오히려 침투 전략으로 식작용을 이용할 수 있다. However, some [[bacteria]] can exploit phagocytosis as an invasion strategy. They either reproduce inside of the phagolysosome ''(e.g. [[Coxiella burnetii|Coxiella]]'' spp.)<ref>{{cite journal | vauthors = Hackstadt T, Williams JC | title = Biochemical stratagem for obligate parasitism of eukaryotic cells by Coxiella burnetii | journal = Proceedings of the National Academy of Sciences of the United States of America | volume = 78 | issue = 5 | pages = 3240–4 | date = May 1981 | pmid = 6942430 | pmc = 319537 | doi = 10.1073/pnas.78.5.3240 | doi-access = free }}</ref> or escape into the [[cytoplasm]] before the phagosome fuses with the lysosome (e.g. ''[[Rickettsia]]'' spp.).<ref>{{cite journal | vauthors = Winkler HH | year = 1990| title = Rickettsia Species (As Organisms) | journal = Annual Review of Microbiology | volume = 44 | pages = 131–153 | doi=10.1146/annurev.micro.44.1.131| pmid = 2252380}}</ref> |
|||
그러나 일부 [[세균]]은 오히려 침투 전략으로 식작용을 이용할 수 있다. 그들은 phagolysosome 내부에서 번식합니다 ''(예:'' ''Coxiella'' spp.) <ref>{{저널 인용|제목=Biochemical stratagem for obligate parasitism of eukaryotic cells by Coxiella burnetii|저널=Proceedings of the National Academy of Sciences of the United States of America|날짜=May 1981|권=78|호=5|쪽=3240–4|doi=10.1073/pnas.78.5.3240|pmc=319537|pmid=6942430}}</ref> phagosome이 lysosome과 융합하기 전에 [[세포질]] 로 탈출(예: ''[[리케차]]'' 종 ). <ref>{{저널 인용|제목=Rickettsia Species (As Organisms)|저널=Annual Review of Microbiology|연도=1990|권=44|쪽=131–153|doi=10.1146/annurev.micro.44.1.131|pmid=2252380}}</ref> ''[[결핵균|Mycobacterium tuberculosis]]'' <ref>{{저널 인용|제목=Immune control of tuberculosis by IFN-gamma-inducible LRG-47|저널=Science|날짜=October 2003|권=302|호=5645|쪽=654–9|bibcode=2003Sci...302..654M|doi=10.1126/science.1088063|pmid=14576437}}</ref> <ref>{{저널 인용|제목=A membrane protein preserves intrabacterial pH in intraphagosomal Mycobacterium tuberculosis|저널=Nature Medicine|날짜=August 2008|권=14|호=8|쪽=849–54|doi=10.1038/nm.1795|pmc=2538620|pmid=18641659}}</ref> 와 ''Mycobacterium avium paratuberculosis'' <ref>{{저널 인용|제목=Characterization of the intracellular survival of Mycobacterium avium ssp. paratuberculosis: phagosomal pH and fusogenicity in J774 macrophages compared with other mycobacteria|저널=Cellular Microbiology|날짜=August 2001|권=3|호=8|쪽=551–66|doi=10.1046/j.1462-5822.2001.00139.x|pmid=11488816}}</ref> 를 포함한 많은 Mycobacteria는 숙주 [[대식세포]] 를 조작하여 리소좀이 phagosome과 융합하여 성숙한 phagolysosome을 생성하는 것을 방지할 수 있습니다. phagosome의 이러한 불완전한 성숙은 내부의 병원체에게 유리한 환경을 유지합니다. <ref>{{저널 인용|제목=How does Mycobacterium avium subsp. paratuberculosis resist intracellular degradation?|저널=The Veterinary Quarterly|날짜=November 2001|권=23|호=4|쪽=153–62|doi=10.1080/01652176.2001.9695105|pmid=11765232}}</ref> |
|||
Many Mycobacteria, including ''[[Mycobacterium tuberculosis]]''<ref>{{cite journal | vauthors = MacMicking JD, Taylor GA, McKinney JD | title = Immune control of tuberculosis by IFN-gamma-inducible LRG-47 | journal = Science | volume = 302 | issue = 5645 | pages = 654–9 | date = October 2003 | pmid = 14576437 | doi = 10.1126/science.1088063 | bibcode = 2003Sci...302..654M | s2cid = 83944695 }}</ref> |
|||
<ref>{{cite journal | vauthors = Vandal OH, Pierini LM, Schnappinger D, Nathan CF, Ehrt S | title = A membrane protein preserves intrabacterial pH in intraphagosomal Mycobacterium tuberculosis | journal = Nature Medicine | volume = 14 | issue = 8 | pages = 849–54 | date = August 2008 | pmid = 18641659 | pmc = 2538620 | doi = 10.1038/nm.1795 }}</ref> |
|||
and ''[[Mycobacterium avium paratuberculosis]]'' |
|||
,<ref>{{cite journal | vauthors = Kuehnel MP, Goethe R, Habermann A, Mueller E, Rohde M, Griffiths G, Valentin-Weigand P | title = Characterization of the intracellular survival of Mycobacterium avium ssp. paratuberculosis: phagosomal pH and fusogenicity in J774 macrophages compared with other mycobacteria | journal = Cellular Microbiology | volume = 3 | issue = 8 | pages = 551–66 | date = August 2001 | pmid = 11488816 | doi = 10.1046/j.1462-5822.2001.00139.x | s2cid = 8962102 }}</ref> can manipulate the host [[macrophage]] to prevent lysosomes from fusing with phagosomes and creating mature phagolysosomes. Such incomplete maturation of the phagosome maintains an environment favorable to the pathogens inside it |
|||
.<ref>{{cite journal | vauthors = Tessema MZ, Koets AP, Rutten VP, Gruys E | title = How does Mycobacterium avium subsp. paratuberculosis resist intracellular degradation? | journal = The Veterinary Quarterly | volume = 23 | issue = 4 | pages = 153–62 | date = November 2001 | pmid = 11765232 | doi = 10.1080/01652176.2001.9695105 | doi-access = free }}</ref> |
|||
==Formation== |
|||
{{further|phagocytosis}} |
|||
Phagosomes are large enough to degrade whole bacteria, or [[apoptosis|apoptotic]] and senescent cells, which are usually >0.5μm in diameter.<ref name="Aderem1999">{{cite journal | vauthors = Aderem A, Underhill DM | title = Mechanisms of phagocytosis in macrophages | journal = Annual Review of Immunology | volume = 17 | issue = 1 | pages = 593–623 | date = April 1999 | pmid = 10358769 | doi = 10.1146/annurev.immunol.17.1.593 }}</ref> This means a phagosome is several orders of magnitude bigger than an [[endosome]], which is measured in [[nanometres]]. |
|||
Phagosomes are formed when pathogens or [[opsonins]] bind to a transmembrane receptor, which are randomly distributed on the phagocyte cell surface. Upon binding, "outside-in" signalling triggers [[actin]] polymerisation and [[pseudopodia]] formation, which surrounds and fuses behind the microorganism. [[Protein kinase C]], [[phosphoinositide 3-kinase]], and [[phospholipase C]] (PLC) are all needed for signalling and controlling particle internalisation.<ref name="Aderem2003" /> More cell surface receptors can bind to the particle in a zipper-like mechanism as the pathogen is surrounded, increasing the binding [[avidity]].<ref name="Dupuy2008">{{cite journal | vauthors = Dupuy AG, Caron E | title = Integrin-dependent phagocytosis: spreading from microadhesion to new concepts | journal = Journal of Cell Science | volume = 121 | issue = 11 | pages = 1773–83 | date = June 2008 | pmid = 18492791 | doi = 10.1242/jcs.018036 | doi-access = free }}</ref> [[Fc receptor]] (FcR), [[complement receptors]] (CR), [[mannose receptor]] and [[dectin-1]] are phagocytic receptors, which means that they can induce phagocytosis if they are expressed in non-phagocytic cells such as [[fibroblasts]].<ref name="Underhill2002">{{cite journal | vauthors = Underhill DM, Ozinsky A | title = Phagocytosis of microbes: complexity in action | journal = Annual Review of Immunology | volume = 20 | issue = 1 | pages = 825–52 | date = April 2002 | pmid = 11861619 | doi = 10.1146/annurev.immunol.20.103001.114744 }}</ref> Other proteins such as [[Toll-like receptors]] are involved in pathogen pattern recognition and are often recruited to phagosomes but do not specifically trigger phagocytosis in non-phagocytic cells, so they are not considered phagocytic receptors. |
|||
===Opsonisation=== |
|||
Opsonins are molecular tags such as [[antibodies]] and [[complement system|complements]] that attach to pathogens and up-regulate phagocytosis. [[Immunoglobulin G]] (IgG) is the major type of antibody present in the [[serum (blood)|serum]]. It is part of the [[adaptive immune system]], but it links to the [[innate immune system|innate response]] by recruiting macrophages to phagocytose pathogens. The antibody binds to microbes with the variable [[Fab domain]], and the [[Fc domain]] binds to Fc receptors (FcR) to induce phagocytosis. |
|||
Complement-mediated internalisation has much less significant membrane protrusions, but the downstream signalling of both pathways converge to activate [[Rho family of GTPases|Rho GTPases]].<ref name="Kaplan1977">{{cite journal | vauthors = Kaplan G | title = Differences in the mode of phagocytosis with Fc and C3 receptors in macrophages | journal = Scandinavian Journal of Immunology | volume = 6 | issue = 8 | pages = 797–807 | date = August 1977 | pmid = 561436 | doi = 10.1111/j.1365-3083.1977.tb02153.x | s2cid = 25373560 }}</ref> They control actin polymerisation which is required for the phagosome to fuse with endosomes and lysosomes. |
|||
===Non-phagocytic cells=== |
|||
Other non-professional phagocytes have some degree of phagocytic activity, such as thyroid and bladder epithelial cells that can engulf erythrocytes and retinal epithelial cells that internalise retinal rods.<ref name="Aderem1999" /> However non-professional phagocytes do not express specific phagocytic receptors such as FcR and have a much lower rate of internalisation. |
|||
Some invasive bacteria can also induce phagocytosis in non-phagocytic cells to mediate host uptake. For example, ''[[Shigella]]'' can secrete toxins that alter the host cytoskeleton and enter the basolateral side of [[enterocytes]].<ref name="Kohler2002">{{cite journal | vauthors = Köhler H, Rodrigues SP, McCormick BA | title = Shigella flexneri Interactions with the Basolateral Membrane Domain of Polarized Model Intestinal Epithelium: Role of Lipopolysaccharide in Cell Invasion and in Activation of the Mitogen-Activated Protein Kinase ERK | journal = Infection and Immunity | volume = 70 | issue = 3 | pages = 1150–8 | date = March 2002 | pmid = 11854195 | pmc = 127798 | doi = 10.1128/IAI.70.3.1150-1158.2002 }}</ref> |
|||
== Structure == |
|||
As the membrane of the phagosome is formed by the fusion of the plasma membrane, the basic composition of the [[phospholipid bilayer]] is the same. Endosomes and lysosomes then fuse with the phagosome to contribute to the membrane, especially when the engulfed particle is very big, such as a [[parasite]].<ref name="Desjardins2005">{{cite journal | vauthors = Desjardins M, Houde M, Gagnon E | title = Phagocytosis: the convoluted way from nutrition to adaptive immunity | journal = Immunological Reviews | volume = 207 | issue = 1 | pages = 158–65 | date = October 2005 | pmid = 16181334 | doi = 10.1111/j.0105-2896.2005.00319.x | s2cid = 30191200 }}</ref> They also deliver various membrane proteins to the phagosome and modify the organelle structure. |
|||
Phagosomes can engulf artificial low-density [[latex]] beads and then purified along a [[sucrose]] concentration gradient, allowing the structure and composition to be studied.<ref name="Desjardins1994" /> By purifying phagosomes at different time points, the maturation process can also be characterised. Early phagosomes are characterised by Rab5, which transition into Rab7 as the vesicle matures into late phagosomes. |
|||
==Maturation process== |
|||
The nascent phagosome is not inherently bactericidal. As it matures, it becomes more acidic from pH 6.5 to pH 4, and gains characteristic protein markers and hydrolytic enzymes. The different enzymes function at various optimal pH, forming a range so they each work in narrow stages of the maturation process. Enzyme activity can be fine-tuned by modifying the pH level, allowing for greater flexibility. The phagosome moves along [[microtubules]] of the [[cytoskeleton]], fusing with endosomes and lysosomes sequentially in a dynamic [[kiss-and-run fusion|"kiss-and-run"]] manner.<ref name="Gotthardt2002" /> This intracellular transport depends on the size of the phagosomes. Larger organelles (with a diameter of about 3 μm) are transported very persistently from the cell periphery towards the perinuclear region whereas smaller organelles (with a diameter of about 1 μm) are transported more bidirectionally back and forth between cell center and cell periphery.<ref name="Keller2017">{{cite journal | vauthors = Keller S, Berghoff K, Kress H | title = Phagosomal transport depends strongly on phagosome size | journal = Scientific Reports | volume = 7 | issue = 1 | pages = 17068 | date = December 2017 | pmid = 29213131 | pmc = 5719076 | doi = 10.1038/s41598-017-17183-7 | bibcode = 2017NatSR...717068K }}</ref> [[v-ATPase|Vacuolar proton pumps]] (v-ATPase) are delivered to the phagosome to acidify the organelle compartment, creating a more hostile environment for pathogens and facilitating protein degradation. The bacterial proteins are denatured in low pH and become more accessible to the proteases, which are unaffected by the acidic environment. The enzymes are later recycled from the phagolysosome before egestion so they are not wasted. The composition of the phospholipid membrane also changes as the phagosome matures.<ref name="Desjardins1994">{{cite journal | vauthors = Desjardins M, Celis JE, van Meer G, Dieplinger H, Jahraus A, Griffiths G, Huber LA | title = Molecular characterization of phagosomes | journal = The Journal of Biological Chemistry | volume = 269 | issue = 51 | pages = 32194–200 | date = December 1994 | doi = 10.1016/S0021-9258(18)31620-X | pmid = 7798218 | doi-access = free }}</ref> |
|||
Fusion may take minutes to hours depending on the contents of the phagosome; FcR or mannose receptor-mediated fusion last less than 30 minutes, but phagosomes containing latex beads may take several hours to fuse with lysosomes.<ref name="Aderem1999" /> It is suggested that the composition of the phagosome membrane affects the rate of maturation. ''Mycobacterium tuberculosis'' has a very hydrophobic [[cell wall]], which is hypothesised to prevent membrane recycling and recruitment of fusion factors, so the phagosome does not fuse with lysosomes and the bacterium avoids degradation.<ref>{{cite journal | vauthors = de Chastellier C, Thilo L | title = Phagosome maturation and fusion with lysosomes in relation to surface property and size of the phagocytic particle | journal = European Journal of Cell Biology | volume = 74 | issue = 1 | pages = 49–62 | date = September 1997 | pmid = 9309390 }}</ref> |
|||
Smaller lumenal molecules are transferred by fusion faster than larger molecules, which suggests that a small aqueous channel forms between the phagosome and other vesicles during "kiss-and-run", through which only limited exchange is allowed.<ref name="Aderem1999" /> |
|||
===Fusion regulation=== |
|||
Shortly after internalisation, F-actin depolymerises from the newly formed phagosome so it becomes accessible to endosomes for fusion and delivery of proteins.<ref name="Aderem1999" /> The maturation process is divided into early and late stages depending on characteristic protein markers, regulated by small Rab GTPases. Rab5 is present on early phagosomes, and controls the transition to late phagosomes marked by Rab7.<ref name="Fairn2012">{{cite journal | vauthors = Fairn GD, Grinstein S | title = How nascent phagosomes mature to become phagolysosomes | journal = Trends in Immunology | volume = 33 | issue = 8 | pages = 397–405 | date = August 2012 | pmid = 22560866 | doi = 10.1016/j.it.2012.03.003 }}</ref> |
|||
Rab5 recruits PI-3 kinase and other tethering proteins such as Vps34 to the phagosome membrane, so endosomes can deliver proteins to the phagosome. Rab5 is partially involved in the transition to Rab7, via the CORVET complex and the HOPS complex in yeast.<ref name="Fairn2012" /> The exact maturation pathway in mammals is not well understood, but it is suggested that HOPS can bind Rab7 and displace the [[guanosine nucleotide dissociation inhibitor]] (GDI).<ref name="Kinchen2010">{{cite journal | vauthors = Kinchen JM, Ravichandran KS | title = Identification of two evolutionarily conserved genes regulating processing of engulfed apoptotic cells | journal = Nature | volume = 464 | issue = 7289 | pages = 778–82 | date = April 2010 | pmid = 20305638 | pmc = 2901565 | doi = 10.1038/nature08853 | bibcode = 2010Natur.464..778K }}</ref> Rab11 is involved in membrane recycling.<ref name="Savina2007" /> |
|||
===Phagolysosome=== |
|||
The phagosome fuses with lysosomes to form a phagolysosome, which has various bactericidal properties. The phagolysosome contains reactive oxygen and [[reactive nitrogen species|nitrogen species]] (ROS and RNS) and hydrolytic enzymes. The compartment is also acidic due to proton pumps (v-ATPases) that transport H<sup>+</sup> across the membrane, used to denature the bacterial proteins. |
|||
The exact properties of phagolysosomes vary depending on the type of phagocyte. Those in dendritic cells have weaker bactericidal properties than those in macrophages and neutrophils. Also, macrophages are divided into pro-inflammatory "killer" M1 and "repair" M2. The phagolysosomes of M1 can metabolise [[arginine]] into highly reactive [[nitric oxide]], while M2 use arginine to produce [[ornithine]] to promote cell proliferation and tissue repair.<ref name="Mills2015">{{cite journal | vauthors = Mills CD | title = Anatomy of a discovery: m1 and m2 macrophages | journal = Frontiers in Immunology | volume = 6 | pages = 212 | date = 5 May 2015 | pmid = 25999950 | pmc = 4419847 | doi = 10.3389/fimmu.2015.00212 | doi-access = free }}</ref> |
|||
== Function == |
|||
===Pathogen degradation=== |
|||
Macrophages and neutrophils are professional phagocytes in charge of most of the pathogen degradation, but they have different bactericidal methods. Neutrophils have granules that fuse with the phagosome. The granules contain [[NADPH oxidase]] and [[myeloperoxidase]], which produce toxic oxygen and chlorine derivatives to kill pathogens in an [[oxidative burst]]. Proteases and [[anti-microbial peptides]] are also released into the phagolysosome. Macrophages lack granules, and rely more on phagolysosome acidification, [[glycosidases]], and proteases to digest microbes.<ref name="Savina2007" /> Phagosomes in dendritic cells are less acidic and have much weaker hydrolytic activity, due to a lower concentration of lysosomal proteases and even the presence of protease inhibitors. |
|||
===Inflammation=== |
|||
Phagosome formation is tied to [[inflammation]] via common signalling molecules. PI-3 kinase and PLC are involved in both the internalisation mechanism and triggering inflammation.<ref name="Aderem2003" /> The two proteins, along with Rho GTPases, are important components of the innate immune response, inducing [[cytokine]] production and activating the [[MAPK/ERK pathway|MAP kinase]] signalling cascade. Pro-inflammatory cytokines including [[interleukin 1 beta|IL-1β]], [[Interleukin 6|IL-6]], [[TNFα]], and [[Ilyushin Il-12|IL-12]] are all produced.<ref name="Aderem1999" /> |
|||
The process is tightly regulated and the inflammatory response varies depending on the particle type within the phagosome. Pathogen-infected apoptotic cells will trigger inflammation, but damaged cells that are degraded as part of the normal tissue turnover do not. The response also differs according to the opsonin-mediated phagocytosis. FcR and mannose receptor-mediated reactions produce pro-inflammatory reactive oxygen species and [[arachidonic acid]] molecules, but CR-mediated reactions do not result in those products.<ref name="Aderem1999" /> |
|||
===Antigen presentation=== |
|||
Immature dendritic cells (DCs) can phagocytose, but mature DCs cannot due to changes in Rho GTPases involved in cytoskeleton remodelling.<ref name="Savina2007" /> The phagosomes of DCs are less hydrolytic and acidic than those of macrophages and neutrophils, as DCs are mainly involved in [[antigen presentation]] rather than pathogen degradation. They need to retain protein fragments of a suitable size for specific bacterial recognition, so the peptides are only partially degraded.<ref name="Savina2007">{{cite journal | vauthors = Savina A, Amigorena S | title = Phagocytosis and antigen presentation in dendritic cells | journal = Immunological Reviews | volume = 219 | issue = 1 | pages = 143–56 | date = October 2007 | pmid = 17850487 | doi = 10.1111/j.1600-065X.2007.00552.x | s2cid = 1267478 }}</ref> Peptides from the bacteria are trafficked to the [[Major Histocompatibility Complex]] (MHC). The peptide antigens are presented to [[lymphocytes]], where they bind to [[T-cell receptors]] and activates [[T-cells]], bridging the gap between innate and adaptive immunity.<ref name="Aderem2003">{{cite journal | vauthors = Aderem A | title = Phagocytosis and the inflammatory response | journal = The Journal of Infectious Diseases | volume = 187 Suppl 2 | issue = s2 | pages = S340-5 | date = June 2003 | pmid = 12792849 | doi = 10.1086/374747 | doi-access = free }}</ref> This is specific to [[mammals]], [[birds]], and jawed fish, as [[insects]] do not have adaptive immunity.<ref name="Warr1997">{{cite journal | vauthors = Warr GW | title = The adaptive immune system of fish | journal = Developments in Biological Standardization | volume = 90 | pages = 15–21 | date = 1997 | pmid = 9270830 }}</ref> |
|||
[[File:Phagocytosis -- amoeba.jpg|thumb|Phagocytosis -- amoeba]] |
|||
===Nutrient=== |
|||
Ancient single-celled organisms such as [[amoeba]] use phagocytosis as a way to acquire nutrients, rather than an immune strategy. They engulf other smaller microbes and digest them within the phagosome of around one bacterium per minute, which is much faster than professional phagocytes.<ref name="Cosson2008">{{cite journal | vauthors = Cosson P, Soldati T | title = Eat, kill or die: when amoeba meets bacteria | journal = Current Opinion in Microbiology | volume = 11 | issue = 3 | pages = 271–6 | date = June 2008 | pmid = 18550419 | doi = 10.1016/j.mib.2008.05.005 }}</ref> For the soil amoeba ''[[Dictyostelium discoideum]]'', their main food source is the bacteria ''[[Legionella pneumophila]]'', which causes [[Legionnaire's disease]] in humans.<ref name="Solomon2000">{{cite journal | vauthors = Solomon JM, Rupper A, Cardelli JA, Isberg RR | title = Intracellular growth of Legionella pneumophila in Dictyostelium discoideum, a system for genetic analysis of host-pathogen interactions | journal = Infection and Immunity | volume = 68 | issue = 5 | pages = 2939–47 | date = May 2000 | pmid = 10768992 | pmc = 97507 | doi = 10.1128/iai.68.5.2939-2947.2000 }}</ref> Phagosome maturation in amoeba is very similar to that in macrophages, so they are used as a model organism to study the process.<ref name="Gotthardt2002">{{cite journal | vauthors = Gotthardt D, Warnatz HJ, Henschel O, Brückert F, Schleicher M, Soldati T | title = High-resolution dissection of phagosome maturation reveals distinct membrane trafficking phases | journal = Molecular Biology of the Cell | volume = 13 | issue = 10 | pages = 3508–20 | date = October 2002 | pmid = 12388753 | pmc = 129962 | doi = 10.1091/mbc.E02-04-0206 }}</ref> |
|||
===Tissue clearance=== |
|||
Phagosomes degrade senescent cells and apoptotic cells to maintain tissue homeostasis. [[Erythrocytes]] have one of the highest turnover rates in the body, and they are phagocytosed by macrophages in the [[liver]] and [[spleen]]. In the [[embryo]], the process of removing dead cells is not well-characterised, but it is not performed by macrophages or other cells derived from [[hematopoietic stem cells]].<ref name="Lichanska2000">{{cite journal | vauthors = Lichanska AM, Hume DA | title = Origins and functions of phagocytes in the embryo | journal = Experimental Hematology | volume = 28 | issue = 6 | pages = 601–11 | date = June 2000 | pmid = 10880746 | doi = 10.1016/s0301-472x(00)00157-0 | url = https://www.pure.ed.ac.uk/ws/files/12628290/Origins_and_functions_of_phagocytes_in_the_embryo.pdf | hdl = 20.500.11820/b865effa-680c-4f60-905e-ba9137f57613 | hdl-access = free }}</ref> It is only in the adult that apoptotic cells are phagocytosed by professional phagocytes. Inflammation is only triggered by certain [[Pathogen-associated molecular pattern|pathogen-]] or [[damage-associated molecular patterns]] (PAMPs or DAMPs), the removal of senescent cells is non-inflammatory.<ref name="Desjardins2005" /> |
|||
===Autophagosome=== |
|||
{{further|Autophagy}} |
|||
[[Autophagosomes]] are different from phagosomes in that they are mainly used to selectively degrade damaged cytosolic organelles such as [[mitochondria]] ([[mitophagy]]). However, when the cell is starved or stressed, autophagosomes can also non-selectively degrade organelles to provide the cell with amino acids and other nutrients.<ref name="Ding2012">{{cite journal | vauthors = Ding WX, Yin XM | title = Mitophagy: mechanisms, pathophysiological roles, and analysis | journal = Biological Chemistry | volume = 393 | issue = 7 | pages = 547–64 | date = July 2012 | pmid = 22944659 | pmc = 3630798 | doi = 10.1515/hsz-2012-0119 }}</ref> Autophagy is not limited to professional phagocytes, it is first discovered in rat [[hepatocytes]] by cell biologist [[Christian de Duve]].<ref>{{cite journal | first = Susana | last = Castro-Obregon | name-list-style = vanc |year= 2010 |title= The Discovery of Lysosomes and Autophagy |url= http://www.nature.com/scitable/topicpage/the-discovery-of-lysosomes-and-autophagy-14199828|journal= Nature Education |volume=3 |issue= 9 | pages=49 }}</ref> Autophagosomes have a double membrane, the inner one from the engulfed organelle, and the outer membrane is speculated to be formed from the [[endoplasmic reticulum]] or the [[Vesicular-tubular cluster|ER-Golgi Intermediate Compartment]] (ERGIC).<ref name="Ge2013">{{cite journal | vauthors = Ge L, Schekman R | title = The ER-Golgi intermediate compartment feeds the phagophore membrane | journal = Autophagy | volume = 10 | issue = 1 | pages = 170–2 | date = January 2014 | pmid = 24220263 | pmc = 4389871 | doi = 10.4161/auto.26787 }}</ref> The autophagosome also fuses with lysosomes to degrade its contents. When ''M. tuberculosis'' inhibit phagosome acidification, [[Interferon gamma]] can induce autophagy and rescue the maturation process.<ref>{{cite journal | vauthors = Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V | title = Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages | journal = Cell | volume = 119 | issue = 6 | pages = 753–66 | date = December 2004 | pmid = 15607973 | doi = 10.1016/j.cell.2004.11.038 | doi-access = free }}</ref> |
|||
==Bacterial evasion and manipulation== |
|||
Many bacteria have evolved to evade the bactericidal properties of phagosomes or even exploit phagocytosis as an invasion strategy. |
|||
*''Mycobacterium tuberculosis'' target M2 macrophages at the lower parts of the respiratory pathway, which do not produce ROS.<ref>{{cite journal | vauthors = Cambier CJ, Takaki KK, Larson RP, Hernandez RE, Tobin DM, Urdahl KB, Cosma CL, Ramakrishnan L | title = Mycobacteria manipulate macrophage recruitment through coordinated use of membrane lipids | journal = Nature | volume = 505 | issue = 7482 | pages = 218–22 | date = January 2014 | pmid = 24336213 | pmc = 3961847 | doi = 10.1038/nature12799 }}</ref> ''M. tuberculosis'' can also manipulate the signalling pathways by secreting phosphatases such as PtpA and SapM, which disrupt protein recruitment and block phagosome acidification.<ref name="Aderem1999" /><ref>{{cite journal | vauthors = Wong D, Chao JD, Av-Gay Y | title = Mycobacterium tuberculosis-secreted phosphatases: from pathogenesis to targets for TB drug development | journal = Trends in Microbiology | volume = 21 | issue = 2 | pages = 100–9 | date = February 2013 | pmid = 23084287 | doi = 10.1016/j.tim.2012.09.002 }}</ref> |
|||
*''Legionella pneumophila'' can re-model the phagosome membrane to imitate vesicles in other parts of the secretory pathway, so lysosomes do not recognise the phagosome and do not fuse with it. The bacterium secretes toxins that interfere with host trafficking, so the ''Legionella''-containing vacuole recruits membrane proteins usually found on the endoplasmic reticulum or the ERGIC.<ref>{{Cite book|last1=Roy|first1=Craig R.|last2=Kagan|first2=Jonathan C. | name-list-style = vanc |title=Evasion of Phagosome Lysosome Fusion and Establishment of a Replicative Organelle by the Intracellular Pathogen Legionella pneumophila|url=https://www.ncbi.nlm.nih.gov/books/NBK6111/|publisher=Landes Bioscience|language=en|date=1 January 2013}}</ref> This re-directs secretory vesicles to the modified phagosome and deliver nutrients to the bacterium. |
|||
*''[[Listeria monocytogenes]]'' secretes a pore-forming protein [[listeriolysin O]] so the bacterium can escape the phagosome into the cytosol. Listeriolysin is activated by the acidic environment of the phagosome.<ref name="Portnoy2002">{{cite journal | vauthors = Portnoy DA, Auerbuch V, Glomski IJ | title = The cell biology of Listeria monocytogenes infection: the intersection of bacterial pathogenesis and cell-mediated immunity | journal = The Journal of Cell Biology | volume = 158 | issue = 3 | pages = 409–14 | date = August 2002 | pmid = 12163465 | pmc = 2173830 | doi = 10.1083/jcb.200205009 }}</ref> In addition, ''Listeria'' secrete two phospholipase C enzymes that facilitate in phagosome escape. |
|||
== See also == |
|||
*[[Autophagosome]] |
|||
*[[Phagocyte]] |
|||
== References == |
|||
{{Reflist|2}} |
|||
{{세포소기관}} |
|||
[[분류:세포생물학]] |
[[분류:세포생물학]] |
||
2022년 4월 23일 (토) 17:42 판
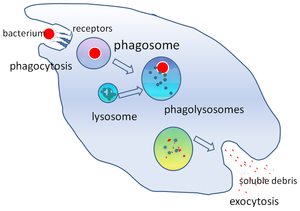
세포생물학에서 파고솜(phagosome)은 식세포가 식작용을 통해 섭취한 입자 주위에 만들어지는 소포이다. 식작용 능력이 뛰어난 전문 식세포에는 대식세포, 호중구, 수지상세포(DC)가 있다.[1]
파고솜은 미생물, 노화된 세포, 세포자멸사 예정인 세포 주변의 세포막이 융합하여 형성된다. 파고솜은 리소좀을 동원하고 서로 융합하여 성숙한 파고리소좀을 형성하는 막 단백질을 가지고 있다. 리소좀에는 병원균을 죽이고 소화시키는 가수 분해 효소와 활성 산소종(ROS)이 포함되어 있다. 파고솜은 식작용 능력이 전문 식세포에 비해서 떨어지는 비전문 식세포에서도 형성될 수 있지만, 더 작은 범위의 입자만 삼킬 수 있으며 ROS는 들어 있지 않다. 소화된 입자에서 유용한 물질(아미노산 등)은 세포질로 이동하고 노폐물은 세포외 배출에 의해 제거된다. 파고솜 형성은 조직 항상성과 병원체에 대한 선천성, 적응성 면역에 중요하다.
그러나 일부 세균은 오히려 침투 전략으로 식작용을 이용할 수 있다. However, some bacteria can exploit phagocytosis as an invasion strategy. They either reproduce inside of the phagolysosome (e.g. Coxiella spp.)[2] or escape into the cytoplasm before the phagosome fuses with the lysosome (e.g. Rickettsia spp.).[3] Many Mycobacteria, including Mycobacterium tuberculosis[4] [5] and Mycobacterium avium paratuberculosis ,[6] can manipulate the host macrophage to prevent lysosomes from fusing with phagosomes and creating mature phagolysosomes. Such incomplete maturation of the phagosome maintains an environment favorable to the pathogens inside it .[7]
Formation
Phagosomes are large enough to degrade whole bacteria, or apoptotic and senescent cells, which are usually >0.5μm in diameter.[8] This means a phagosome is several orders of magnitude bigger than an endosome, which is measured in nanometres.
Phagosomes are formed when pathogens or opsonins bind to a transmembrane receptor, which are randomly distributed on the phagocyte cell surface. Upon binding, "outside-in" signalling triggers actin polymerisation and pseudopodia formation, which surrounds and fuses behind the microorganism. Protein kinase C, phosphoinositide 3-kinase, and phospholipase C (PLC) are all needed for signalling and controlling particle internalisation.[9] More cell surface receptors can bind to the particle in a zipper-like mechanism as the pathogen is surrounded, increasing the binding avidity.[10] Fc receptor (FcR), complement receptors (CR), mannose receptor and dectin-1 are phagocytic receptors, which means that they can induce phagocytosis if they are expressed in non-phagocytic cells such as fibroblasts.[11] Other proteins such as Toll-like receptors are involved in pathogen pattern recognition and are often recruited to phagosomes but do not specifically trigger phagocytosis in non-phagocytic cells, so they are not considered phagocytic receptors.
Opsonisation
Opsonins are molecular tags such as antibodies and complements that attach to pathogens and up-regulate phagocytosis. Immunoglobulin G (IgG) is the major type of antibody present in the serum. It is part of the adaptive immune system, but it links to the innate response by recruiting macrophages to phagocytose pathogens. The antibody binds to microbes with the variable Fab domain, and the Fc domain binds to Fc receptors (FcR) to induce phagocytosis.
Complement-mediated internalisation has much less significant membrane protrusions, but the downstream signalling of both pathways converge to activate Rho GTPases.[12] They control actin polymerisation which is required for the phagosome to fuse with endosomes and lysosomes.
Non-phagocytic cells
Other non-professional phagocytes have some degree of phagocytic activity, such as thyroid and bladder epithelial cells that can engulf erythrocytes and retinal epithelial cells that internalise retinal rods.[8] However non-professional phagocytes do not express specific phagocytic receptors such as FcR and have a much lower rate of internalisation.
Some invasive bacteria can also induce phagocytosis in non-phagocytic cells to mediate host uptake. For example, Shigella can secrete toxins that alter the host cytoskeleton and enter the basolateral side of enterocytes.[13]
Structure
As the membrane of the phagosome is formed by the fusion of the plasma membrane, the basic composition of the phospholipid bilayer is the same. Endosomes and lysosomes then fuse with the phagosome to contribute to the membrane, especially when the engulfed particle is very big, such as a parasite.[14] They also deliver various membrane proteins to the phagosome and modify the organelle structure.
Phagosomes can engulf artificial low-density latex beads and then purified along a sucrose concentration gradient, allowing the structure and composition to be studied.[15] By purifying phagosomes at different time points, the maturation process can also be characterised. Early phagosomes are characterised by Rab5, which transition into Rab7 as the vesicle matures into late phagosomes.
Maturation process
The nascent phagosome is not inherently bactericidal. As it matures, it becomes more acidic from pH 6.5 to pH 4, and gains characteristic protein markers and hydrolytic enzymes. The different enzymes function at various optimal pH, forming a range so they each work in narrow stages of the maturation process. Enzyme activity can be fine-tuned by modifying the pH level, allowing for greater flexibility. The phagosome moves along microtubules of the cytoskeleton, fusing with endosomes and lysosomes sequentially in a dynamic "kiss-and-run" manner.[16] This intracellular transport depends on the size of the phagosomes. Larger organelles (with a diameter of about 3 μm) are transported very persistently from the cell periphery towards the perinuclear region whereas smaller organelles (with a diameter of about 1 μm) are transported more bidirectionally back and forth between cell center and cell periphery.[17] Vacuolar proton pumps (v-ATPase) are delivered to the phagosome to acidify the organelle compartment, creating a more hostile environment for pathogens and facilitating protein degradation. The bacterial proteins are denatured in low pH and become more accessible to the proteases, which are unaffected by the acidic environment. The enzymes are later recycled from the phagolysosome before egestion so they are not wasted. The composition of the phospholipid membrane also changes as the phagosome matures.[15]
Fusion may take minutes to hours depending on the contents of the phagosome; FcR or mannose receptor-mediated fusion last less than 30 minutes, but phagosomes containing latex beads may take several hours to fuse with lysosomes.[8] It is suggested that the composition of the phagosome membrane affects the rate of maturation. Mycobacterium tuberculosis has a very hydrophobic cell wall, which is hypothesised to prevent membrane recycling and recruitment of fusion factors, so the phagosome does not fuse with lysosomes and the bacterium avoids degradation.[18]
Smaller lumenal molecules are transferred by fusion faster than larger molecules, which suggests that a small aqueous channel forms between the phagosome and other vesicles during "kiss-and-run", through which only limited exchange is allowed.[8]
Fusion regulation
Shortly after internalisation, F-actin depolymerises from the newly formed phagosome so it becomes accessible to endosomes for fusion and delivery of proteins.[8] The maturation process is divided into early and late stages depending on characteristic protein markers, regulated by small Rab GTPases. Rab5 is present on early phagosomes, and controls the transition to late phagosomes marked by Rab7.[19]
Rab5 recruits PI-3 kinase and other tethering proteins such as Vps34 to the phagosome membrane, so endosomes can deliver proteins to the phagosome. Rab5 is partially involved in the transition to Rab7, via the CORVET complex and the HOPS complex in yeast.[19] The exact maturation pathway in mammals is not well understood, but it is suggested that HOPS can bind Rab7 and displace the guanosine nucleotide dissociation inhibitor (GDI).[20] Rab11 is involved in membrane recycling.[21]
Phagolysosome
The phagosome fuses with lysosomes to form a phagolysosome, which has various bactericidal properties. The phagolysosome contains reactive oxygen and nitrogen species (ROS and RNS) and hydrolytic enzymes. The compartment is also acidic due to proton pumps (v-ATPases) that transport H+ across the membrane, used to denature the bacterial proteins.
The exact properties of phagolysosomes vary depending on the type of phagocyte. Those in dendritic cells have weaker bactericidal properties than those in macrophages and neutrophils. Also, macrophages are divided into pro-inflammatory "killer" M1 and "repair" M2. The phagolysosomes of M1 can metabolise arginine into highly reactive nitric oxide, while M2 use arginine to produce ornithine to promote cell proliferation and tissue repair.[22]
Function
Pathogen degradation
Macrophages and neutrophils are professional phagocytes in charge of most of the pathogen degradation, but they have different bactericidal methods. Neutrophils have granules that fuse with the phagosome. The granules contain NADPH oxidase and myeloperoxidase, which produce toxic oxygen and chlorine derivatives to kill pathogens in an oxidative burst. Proteases and anti-microbial peptides are also released into the phagolysosome. Macrophages lack granules, and rely more on phagolysosome acidification, glycosidases, and proteases to digest microbes.[21] Phagosomes in dendritic cells are less acidic and have much weaker hydrolytic activity, due to a lower concentration of lysosomal proteases and even the presence of protease inhibitors.
Inflammation
Phagosome formation is tied to inflammation via common signalling molecules. PI-3 kinase and PLC are involved in both the internalisation mechanism and triggering inflammation.[9] The two proteins, along with Rho GTPases, are important components of the innate immune response, inducing cytokine production and activating the MAP kinase signalling cascade. Pro-inflammatory cytokines including IL-1β, IL-6, TNFα, and IL-12 are all produced.[8]
The process is tightly regulated and the inflammatory response varies depending on the particle type within the phagosome. Pathogen-infected apoptotic cells will trigger inflammation, but damaged cells that are degraded as part of the normal tissue turnover do not. The response also differs according to the opsonin-mediated phagocytosis. FcR and mannose receptor-mediated reactions produce pro-inflammatory reactive oxygen species and arachidonic acid molecules, but CR-mediated reactions do not result in those products.[8]
Antigen presentation
Immature dendritic cells (DCs) can phagocytose, but mature DCs cannot due to changes in Rho GTPases involved in cytoskeleton remodelling.[21] The phagosomes of DCs are less hydrolytic and acidic than those of macrophages and neutrophils, as DCs are mainly involved in antigen presentation rather than pathogen degradation. They need to retain protein fragments of a suitable size for specific bacterial recognition, so the peptides are only partially degraded.[21] Peptides from the bacteria are trafficked to the Major Histocompatibility Complex (MHC). The peptide antigens are presented to lymphocytes, where they bind to T-cell receptors and activates T-cells, bridging the gap between innate and adaptive immunity.[9] This is specific to mammals, birds, and jawed fish, as insects do not have adaptive immunity.[23]

Nutrient
Ancient single-celled organisms such as amoeba use phagocytosis as a way to acquire nutrients, rather than an immune strategy. They engulf other smaller microbes and digest them within the phagosome of around one bacterium per minute, which is much faster than professional phagocytes.[24] For the soil amoeba Dictyostelium discoideum, their main food source is the bacteria Legionella pneumophila, which causes Legionnaire's disease in humans.[25] Phagosome maturation in amoeba is very similar to that in macrophages, so they are used as a model organism to study the process.[16]
Tissue clearance
Phagosomes degrade senescent cells and apoptotic cells to maintain tissue homeostasis. Erythrocytes have one of the highest turnover rates in the body, and they are phagocytosed by macrophages in the liver and spleen. In the embryo, the process of removing dead cells is not well-characterised, but it is not performed by macrophages or other cells derived from hematopoietic stem cells.[26] It is only in the adult that apoptotic cells are phagocytosed by professional phagocytes. Inflammation is only triggered by certain pathogen- or damage-associated molecular patterns (PAMPs or DAMPs), the removal of senescent cells is non-inflammatory.[14]
Autophagosome
Autophagosomes are different from phagosomes in that they are mainly used to selectively degrade damaged cytosolic organelles such as mitochondria (mitophagy). However, when the cell is starved or stressed, autophagosomes can also non-selectively degrade organelles to provide the cell with amino acids and other nutrients.[27] Autophagy is not limited to professional phagocytes, it is first discovered in rat hepatocytes by cell biologist Christian de Duve.[28] Autophagosomes have a double membrane, the inner one from the engulfed organelle, and the outer membrane is speculated to be formed from the endoplasmic reticulum or the ER-Golgi Intermediate Compartment (ERGIC).[29] The autophagosome also fuses with lysosomes to degrade its contents. When M. tuberculosis inhibit phagosome acidification, Interferon gamma can induce autophagy and rescue the maturation process.[30]
Bacterial evasion and manipulation
Many bacteria have evolved to evade the bactericidal properties of phagosomes or even exploit phagocytosis as an invasion strategy.
- Mycobacterium tuberculosis target M2 macrophages at the lower parts of the respiratory pathway, which do not produce ROS.[31] M. tuberculosis can also manipulate the signalling pathways by secreting phosphatases such as PtpA and SapM, which disrupt protein recruitment and block phagosome acidification.[8][32]
- Legionella pneumophila can re-model the phagosome membrane to imitate vesicles in other parts of the secretory pathway, so lysosomes do not recognise the phagosome and do not fuse with it. The bacterium secretes toxins that interfere with host trafficking, so the Legionella-containing vacuole recruits membrane proteins usually found on the endoplasmic reticulum or the ERGIC.[33] This re-directs secretory vesicles to the modified phagosome and deliver nutrients to the bacterium.
- Listeria monocytogenes secretes a pore-forming protein listeriolysin O so the bacterium can escape the phagosome into the cytosol. Listeriolysin is activated by the acidic environment of the phagosome.[34] In addition, Listeria secrete two phospholipase C enzymes that facilitate in phagosome escape.
See also
References
- ↑ Robinson & Babcock 1998 and Ernst & Stendahl 2006
- ↑ Hackstadt T, Williams JC (May 1981). “Biochemical stratagem for obligate parasitism of eukaryotic cells by Coxiella burnetii”. 《Proceedings of the National Academy of Sciences of the United States of America》 78 (5): 3240–4. doi:10.1073/pnas.78.5.3240. PMC 319537. PMID 6942430.
- ↑ Winkler HH (1990). “Rickettsia Species (As Organisms)”. 《Annual Review of Microbiology》 44: 131–153. doi:10.1146/annurev.micro.44.1.131. PMID 2252380.
- ↑ MacMicking JD, Taylor GA, McKinney JD (October 2003). “Immune control of tuberculosis by IFN-gamma-inducible LRG-47”. 《Science》 302 (5645): 654–9. Bibcode:2003Sci...302..654M. doi:10.1126/science.1088063. PMID 14576437. S2CID 83944695.
- ↑ Vandal OH, Pierini LM, Schnappinger D, Nathan CF, Ehrt S (August 2008). “A membrane protein preserves intrabacterial pH in intraphagosomal Mycobacterium tuberculosis”. 《Nature Medicine》 14 (8): 849–54. doi:10.1038/nm.1795. PMC 2538620. PMID 18641659.
- ↑ Kuehnel MP, Goethe R, Habermann A, Mueller E, Rohde M, Griffiths G, Valentin-Weigand P (August 2001). “Characterization of the intracellular survival of Mycobacterium avium ssp. paratuberculosis: phagosomal pH and fusogenicity in J774 macrophages compared with other mycobacteria”. 《Cellular Microbiology》 3 (8): 551–66. doi:10.1046/j.1462-5822.2001.00139.x. PMID 11488816. S2CID 8962102.
- ↑ Tessema MZ, Koets AP, Rutten VP, Gruys E (November 2001). “How does Mycobacterium avium subsp. paratuberculosis resist intracellular degradation?”. 《The Veterinary Quarterly》 23 (4): 153–62. doi:10.1080/01652176.2001.9695105. PMID 11765232.
- ↑ 가 나 다 라 마 바 사 아 Aderem A, Underhill DM (April 1999). “Mechanisms of phagocytosis in macrophages”. 《Annual Review of Immunology》 17 (1): 593–623. doi:10.1146/annurev.immunol.17.1.593. PMID 10358769.
- ↑ 가 나 다 Aderem A (June 2003). “Phagocytosis and the inflammatory response”. 《The Journal of Infectious Diseases》. 187 Suppl 2 (s2): S340–5. doi:10.1086/374747. PMID 12792849.
- ↑ Dupuy AG, Caron E (June 2008). “Integrin-dependent phagocytosis: spreading from microadhesion to new concepts”. 《Journal of Cell Science》 121 (11): 1773–83. doi:10.1242/jcs.018036. PMID 18492791.
- ↑ Underhill DM, Ozinsky A (April 2002). “Phagocytosis of microbes: complexity in action”. 《Annual Review of Immunology》 20 (1): 825–52. doi:10.1146/annurev.immunol.20.103001.114744. PMID 11861619.
- ↑ Kaplan G (August 1977). “Differences in the mode of phagocytosis with Fc and C3 receptors in macrophages”. 《Scandinavian Journal of Immunology》 6 (8): 797–807. doi:10.1111/j.1365-3083.1977.tb02153.x. PMID 561436. S2CID 25373560.
- ↑ Köhler H, Rodrigues SP, McCormick BA (March 2002). “Shigella flexneri Interactions with the Basolateral Membrane Domain of Polarized Model Intestinal Epithelium: Role of Lipopolysaccharide in Cell Invasion and in Activation of the Mitogen-Activated Protein Kinase ERK”. 《Infection and Immunity》 70 (3): 1150–8. doi:10.1128/IAI.70.3.1150-1158.2002. PMC 127798. PMID 11854195.
- ↑ 가 나 Desjardins M, Houde M, Gagnon E (October 2005). “Phagocytosis: the convoluted way from nutrition to adaptive immunity”. 《Immunological Reviews》 207 (1): 158–65. doi:10.1111/j.0105-2896.2005.00319.x. PMID 16181334. S2CID 30191200.
- ↑ 가 나 Desjardins M, Celis JE, van Meer G, Dieplinger H, Jahraus A, Griffiths G, Huber LA (December 1994). “Molecular characterization of phagosomes”. 《The Journal of Biological Chemistry》 269 (51): 32194–200. doi:10.1016/S0021-9258(18)31620-X. PMID 7798218.
- ↑ 가 나 Gotthardt D, Warnatz HJ, Henschel O, Brückert F, Schleicher M, Soldati T (October 2002). “High-resolution dissection of phagosome maturation reveals distinct membrane trafficking phases”. 《Molecular Biology of the Cell》 13 (10): 3508–20. doi:10.1091/mbc.E02-04-0206. PMC 129962. PMID 12388753.
- ↑ Keller S, Berghoff K, Kress H (December 2017). “Phagosomal transport depends strongly on phagosome size”. 《Scientific Reports》 7 (1): 17068. Bibcode:2017NatSR...717068K. doi:10.1038/s41598-017-17183-7. PMC 5719076. PMID 29213131.
- ↑ de Chastellier C, Thilo L (September 1997). “Phagosome maturation and fusion with lysosomes in relation to surface property and size of the phagocytic particle”. 《European Journal of Cell Biology》 74 (1): 49–62. PMID 9309390.
- ↑ 가 나 Fairn GD, Grinstein S (August 2012). “How nascent phagosomes mature to become phagolysosomes”. 《Trends in Immunology》 33 (8): 397–405. doi:10.1016/j.it.2012.03.003. PMID 22560866.
- ↑ Kinchen JM, Ravichandran KS (April 2010). “Identification of two evolutionarily conserved genes regulating processing of engulfed apoptotic cells”. 《Nature》 464 (7289): 778–82. Bibcode:2010Natur.464..778K. doi:10.1038/nature08853. PMC 2901565. PMID 20305638.
- ↑ 가 나 다 라 Savina A, Amigorena S (October 2007). “Phagocytosis and antigen presentation in dendritic cells”. 《Immunological Reviews》 219 (1): 143–56. doi:10.1111/j.1600-065X.2007.00552.x. PMID 17850487. S2CID 1267478.
- ↑ Mills CD (2015년 5월 5일). “Anatomy of a discovery: m1 and m2 macrophages”. 《Frontiers in Immunology》 6: 212. doi:10.3389/fimmu.2015.00212. PMC 4419847. PMID 25999950.
- ↑ Warr GW (1997). “The adaptive immune system of fish”. 《Developments in Biological Standardization》 90: 15–21. PMID 9270830.
- ↑ Cosson P, Soldati T (June 2008). “Eat, kill or die: when amoeba meets bacteria”. 《Current Opinion in Microbiology》 11 (3): 271–6. doi:10.1016/j.mib.2008.05.005. PMID 18550419.
- ↑ Solomon JM, Rupper A, Cardelli JA, Isberg RR (May 2000). “Intracellular growth of Legionella pneumophila in Dictyostelium discoideum, a system for genetic analysis of host-pathogen interactions”. 《Infection and Immunity》 68 (5): 2939–47. doi:10.1128/iai.68.5.2939-2947.2000. PMC 97507. PMID 10768992.
- ↑ Lichanska AM, Hume DA (June 2000). “Origins and functions of phagocytes in the embryo” (PDF). 《Experimental Hematology》 28 (6): 601–11. doi:10.1016/s0301-472x(00)00157-0. hdl:20.500.11820/b865effa-680c-4f60-905e-ba9137f57613. PMID 10880746.
- ↑ Ding WX, Yin XM (July 2012). “Mitophagy: mechanisms, pathophysiological roles, and analysis”. 《Biological Chemistry》 393 (7): 547–64. doi:10.1515/hsz-2012-0119. PMC 3630798. PMID 22944659.
- ↑ Castro-Obregon, Susana (2010). “The Discovery of Lysosomes and Autophagy”. 《Nature Education》 3 (9): 49.
- ↑ Ge L, Schekman R (January 2014). “The ER-Golgi intermediate compartment feeds the phagophore membrane”. 《Autophagy》 10 (1): 170–2. doi:10.4161/auto.26787. PMC 4389871. PMID 24220263.
- ↑ Gutierrez MG, Master SS, Singh SB, Taylor GA, Colombo MI, Deretic V (December 2004). “Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages”. 《Cell》 119 (6): 753–66. doi:10.1016/j.cell.2004.11.038. PMID 15607973.
- ↑ Cambier CJ, Takaki KK, Larson RP, Hernandez RE, Tobin DM, Urdahl KB, Cosma CL, Ramakrishnan L (January 2014). “Mycobacteria manipulate macrophage recruitment through coordinated use of membrane lipids”. 《Nature》 505 (7482): 218–22. doi:10.1038/nature12799. PMC 3961847. PMID 24336213.
- ↑ Wong D, Chao JD, Av-Gay Y (February 2013). “Mycobacterium tuberculosis-secreted phosphatases: from pathogenesis to targets for TB drug development”. 《Trends in Microbiology》 21 (2): 100–9. doi:10.1016/j.tim.2012.09.002. PMID 23084287.
- ↑ Roy, Craig R.; Kagan, Jonathan C. (2013년 1월 1일). 《Evasion of Phagosome Lysosome Fusion and Establishment of a Replicative Organelle by the Intracellular Pathogen Legionella pneumophila》 (영어). Landes Bioscience.
- ↑ Portnoy DA, Auerbuch V, Glomski IJ (August 2002). “The cell biology of Listeria monocytogenes infection: the intersection of bacterial pathogenesis and cell-mediated immunity”. 《The Journal of Cell Biology》 158 (3): 409–14. doi:10.1083/jcb.200205009. PMC 2173830. PMID 12163465.
